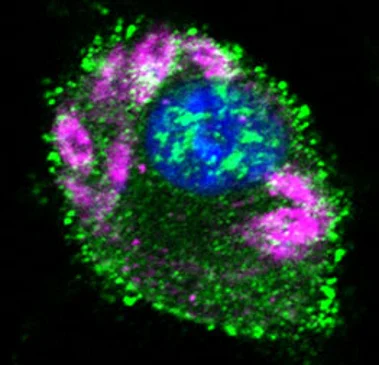
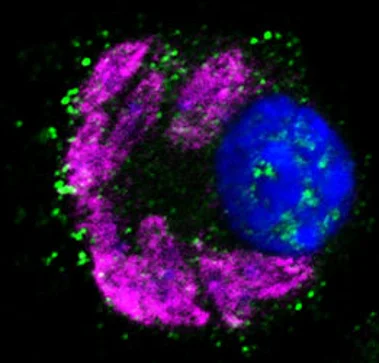
(images: courtesy to Katrin Paduch, Institute of Clinical Microbiology, Immunology and Hygiene, FAU)
Research Topics of the Laboratory for Innate Immunity and Leishmania Research
Leishmania parasites are protozoan pathogens which are transmitted by sand flies to various mammalian hosts, where they cause a broad range of diseases ranging from local and ultimately self-healing lesions to systemic, visceral infections that are lethal, if untreated. Studies with well-established mouse models of leishmaniasis revealed that both the innate and adaptive immune system (macrophages, dendritic cells (DCs), natural killer (NK) cells, CD4+ T helper (Th) cells, CD8+ T cells, B lymphocytes and different cytokines such as IFN-g, TNF and type I inferferons) contribute to an efficient immune response against the intracellular parasites.
The focus of our research work is the analysis of cells, receptors, cytokines and antimicrobial effector mechanisms of the innate immune system during Leishmania infections. Using high resolution imaging techniques, single cell RNAseq analyses, immunophysical methods and transgenic mouse models, we aim to elucidate the immunological processes and the tissue micromilieu that account for the control or evasion of the intracellular protozoan parasite Leishmania (L. major, L. mexicana and L. infantum)and to define novel approaches for the treatment of human leishmaniasis.
In current projects special emphasis is given to the
A) function and regulation of arginase 1, arginase 2 and inducible nitric oxide synthase (iNOS, NOS2) during cutaneous leishmaniasis (L. major and L. mexicana (supported by the German Research Foundation, RTG 2740)
B) molecular mechanisms of TNF-mediated control of Leishmania parasites (supported by the German Research Foundation, BO996/7-1 and SCHL615/3-1)
C) role of natural killer cells and innate lymphoid cells (ILC1, ILC2) during cutaneous (L. major) and visceral infection (L. infantum) (supported by the German Research Foundation, DFG, BO 996/5-2 and SCHL615/2-1)
D) characterization of the function and micromilieu-dependent regulation of macrophages, dendritic cells, eosinophils and mesenchymal cells in L. major and L. mexicana infections (supported by the German Research Foundation, RTG 2740)
E) crossregulation of inducible nitric oxide synthase (iNOS, NOS2) and iron in cutaneous and visceral leishmaniasis
F) therapeutic and immunoregulatory effects of reactive chlorine oxygen species in human cutaneous leishmaniasis (together with Waisenmedizin e.V. Freiburg https://waisenmedizin.org/ )
Recent publications in the field of leishmaniasis
- Rai B, Barinberg D, Chunguang L, Debus A, Rauh M, Kirici S, Sebald H, Grotz M, Gold T, Kunz M, Bozec A, Mattner J, Roberts S, Becker I, Bogdan C*, Schleicher U*. (2025). Oral L-arginine cures arginase 1-dependent chronic cutaneous leishmaniasis by redirecting the T helper cell response. bioRxiv 10.1101/2025.09.23.677696 https://www.biorxiv.org/content/10.1101/2025.09.23.677696v1(*shared senior and corresponding authorship)
- Barinberg D, Sebald H, Gold T, Rai R, Radtke D, Lerm D, Voehringer D, Jantsch J, Wirtz S, Antonova AU, Colonna M, Bogdan C*, Schleicher U*. (2025). The metabolic program of inflammatory eosinophils accounts for chronic parasite-induced skin disease. bioRxiv 2025.02.28.640104; doi: https://doi.org/10.1101/2025.02.28.640104 (*shared senior and corresponding authorship)
- Esse J, Träger J, Steininger P, Bihlmaier K, Fürst J, Bardonicsek-Depnering Z, Naumann-Bartsch N, Morhart P, Castellanos I, Krause SW, Herbst L, Strauß R, Chada M, Korn K, Valenza G, Teschner D, Bogdan C, Held J. (2025). Metagenomic analysis of microbial cell-free DNA from plasma of patients with suspected infections: performance and therapeutic impact in clinical routine.Clin Microbiol Infect. 2025 Jun;31(6):1018-1025. doi: 10.1016/j.cmi.2025.02.016.
- Bogdan C, Islam NA, Barinberg D, Soulat D, Schleicher U, Rai B. (2024). The immunomicrotope of Leishmania control and persistence. Trends Parasitol 40: 788-804. 10.1016/j.pt.2024.07.013.
- Lai C, Heinemann J, Schleicher U, Schett G, Bogdan C, Bozec A, Soulat D. Chronic Systemic Infection of Mice with Leishmania infantum Leads to Increased Bone Mass. J Bone Miner Res. 2023 Jan;38(1):86-102. doi: 10.1002/jbmr.4733. Epub 2022 Nov 16.
- Debus D, Genç S, Kurz P, Holzer M, Bauer K, Heimke-Brinck R, Baier M, Sebald H, Debus A, Bogdan C, Stahl KW. Case Report: Local Treatment of a Leishmania tropica Infection in a Syrian Child with a Novel Filmogenic Preparation of Pharmaceutical Sodium Chlorite (LeiProtect®). Am J Trop Med Hyg. 2022 Jan 10;106(3):857-860. doi: 10.4269/ajtmh.21-0962.
- Sasse C, Barinberg D, Obermeyer S, Debus A, Schleicher U, Bogdan C. Eosinophils, but Not Type 2 Innate Lymphoid Cells, Are the Predominant Source of Interleukin 4 during the Innate Phase of Leishmania major Infection. Pathogens 2022 Jul 25;11(8):828. doi: 10.3390/pathogens11080828.
- Bogdan C.Macrophages as host, effector and immunoregulatory cells in leishmaniasis: Impact of tissue micro-environment and metabolism. Cytokine X. 2020 Oct 12;2(4):100041. doi: 10.1016/j.cytox.2020.100041. eCollection 2020 Dec.
- Paduch K, Debus A, Rai B, Schleicher U, Bogdan C. Resolution of cutaneous leishmaniasis and persistence of Leishmania major in the absence of arginase 1. J Immunol. 2019 Mar 1;202(5):1453-1464. doi: 10.4049/jimmunol.1801249. Epub 2019 Jan 21.
- Bogdan C, Debus A, Sebald H, Rai B, Schäfer J, Obermeyer S, Schleicher U. Experimental Cutaneous Leishmaniasis: Mouse Models for Resolution of Inflammation Versus Chronicity of Disease. Methods Mol Biol. 2019;1971:315-349. doi: 10.1007/978-1-4939-9210-2_18.
- Messlinger H, Sebald H, Heger L, Dudziak D, Bogdan C and Schleicher U. (2018). Monocyte-derived signals activate human natural killer cells in response to Leishmania parasites. Frontiers in Immunology epublished January 24, 2018: doi: 10.3389/fimmu.2018.00024.
- Schleicher U, Liese J, Justies N, Mischke T, Haeberlein S, Sebald H, Kalinke U, Weiss S and Bogdan C. (2018). Type I interferon signaling is required for CpG-oligodesoxy-nucleotide-induced control of Leishmania major, but not for spontaneous cure of subcutaneous cure of primary and secondary L. major infection. Frontiers in Immunology epublished January 11, 2018: doi: 10.3389/ fimmu.2018.00079.
- Schleicher U., Paduch K., Debus A., Obermeyer S., König T., Kling J.C., Ribechini E., Dudziak D., Mougiakakos D., Murray P.J., Ostuni R., Körner H. and Bogdan C (2016). TNF-Mediated Restriction of Arginase 1 Expression in Myeloid Cells Triggers Type 2 NO Synthase Activity at the Site of Infection. Cell Reports 15: 1062-75.
- Bogdan C (2015) Nitric oxide synthase in innate and adaptive immunity: an update. Trends Immunol 36: 161-178
- Schleicher U, Liese J, Knippertz I, Kurzmann C, Hesse A, Heit A, Fischer JA, Weiss S, Kalinke U, Kunz S, Bogdan C (2007). NK cell activation in visceral leishmaniasis requires TLR9, myeloid DCs, and IL-12, but is independent of pDCs. J Exp Med. 204: 893-906.



