Publications from Lang lab
Human macrophages and monocytes need TNF for MINCLE expression, too
Carl Haberkamp, Ida Allabauer, Niklas Blaha, Barbara Bodendorfer, Sarah Cunningham, Andre Hoerning, Roland Lang. 2023. TNF promotes DECTIN2 family C-type lectin receptor expression in human macrophages. Journal of Leukocyte Biology. https://doi.org/10.1093/jleuko/qiad029
MD thesis student Carl Haberkamp's manuscript on regulation of MINCLE by TNF in human macrophages just came out as Advance Article online in Journal of Leukocyte Biology. The results translate our previous findings of an essential role for TNF in upregulation of MINCLE to the human system. They also indicate that blockade of TNF for treatment of inflammatory disease may inhibit expression of these DECTIN2 family pattern recognition receptors in patients.
Congratulations to Carl, and thanks to coauthors Niklas Blaha and Barbara Bodendorfer from our Institute, Ida Allabauer and Andre Hoerning from Department of Pediatrics and Sarah Cunningham from Department of Transfusion Medicine.
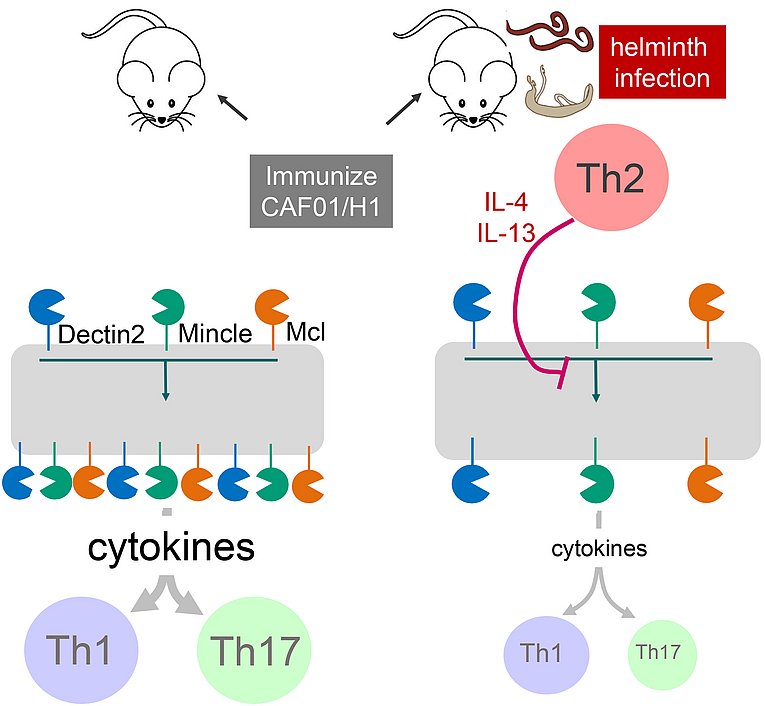
A potential mechanism how worm infection impedes sensing of mycobacteria and vaccine responses
Judith Schick, Meltem Altunay, Matthew Lacorcia, Nathalie Marschner, Stefanie Westermann, Julia Schluckebier, Christoph Schubart, Barbara Bodendorfer, Dennis Christensen, Christian Alexander, Stefan Wirtz, David Voehringer, Clarissa Prazeres da Costa, Roland Lang. 2023. IL-4 and helminth infection downregulate MINCLE-dependent macrophage response to mycobacteria and Th17 adjuvanticity.
eLife. https://doi.org/10.7554/eLife.72923
Epidemiological data show that worm infections can increase the risk to become infected with Mycobacterium tuberculosis and to develop active disease. The C-type lectin receptors (MINCLE, MCL and DECTIN2) recognize defined cell wall components of the mycobacteria causing tuberculosis. In this manuscript published today in eLife we show that the Th2 cytokine IL-4 inhibits the expression of these receptors on macrophages and their response to live mycobacteria. Mice with underlying worm infection expressed less MINCLE receptor on monocytes and were impaired in generating a Th17 response to vaccination with a MINCLE-dependent adjuvant.
These findings implicate negative regulation of CLR expression by IL-4 as a new mechanism of suppressed sensing of the tuberculosis bacillus and of thwarted vaccine responses in patients with underlying worm infections. Former PhD student Judith Schick spearheaded the work, which was completed by current PhD student Meltem Altunay, in collaboration with the research groups of David Voehringer (Dpt. Infection Biology, University Hospital Erlangen) and Clarissa Prazeres da Costa (Medical Microbiology, TU Munich).
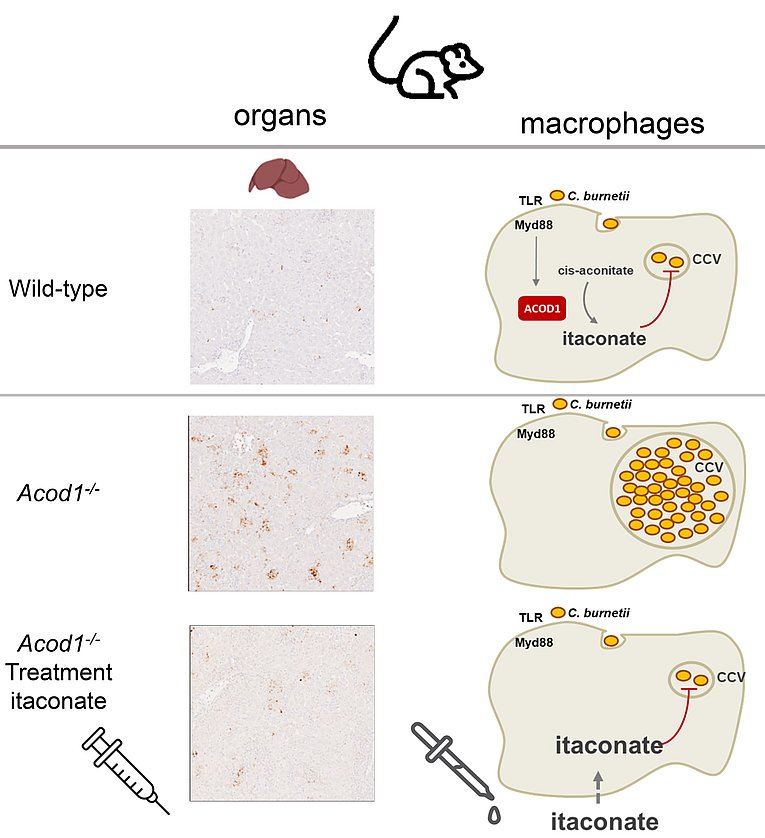
Macrophages produce itaconate to fight the intracellular pathogen Coxiella burnetii
Kohl, L., M. Siddique, B. Bodendorfer, R. Berger, A. Preikschat, C. Daniel, M. Olke, E. Liebler-Tenorio, J. Schulze-Luehrmann, M. Mauermeir, K. T. Yang, I. Hayek, M. Szperlinski, J. Andrack, U. Schleicher, A. Bozec, G. Kronke, P. J. Murray, S. Wirtz, M. Yamamoto, V. Schatz, J. Jantsch, P. Oefner, D. Degrandi, K. Pfeffer, K. Mertens-Scholz, S. Rauber, C. Bogdan, K. Dettmer, A. Luhrmann, and R. Lang. 2022. Macrophages inhibit Coxiella burnetii by the ACOD1-itaconate pathway for containment of Q fever. EMBO Mol Med: e15931.
www.embopress.org/doi/full/10.15252/emmm.202215931
The intracellular bacterium Coxiella burnetii causes the anthropozoonotic infection Q fever. Most patients control and resolve infection, but some develop chronic disease. Finding out how macrophages inhibit bacterial replication may explain susceptibility and suggest new therapies. Lisa Kohl, Md. Nur Siddique and Barbara Bodendorfer have now identified the immunometabolite itaconate as a key factor for the innate host defence by macrophages against Q fever. C. burnetii induces expression of the enzyme Aconitate Decarboxylase 1 (ACOD1) in mouse and human macrophages. Acod1-/- mice and macrophages do not generate the immunometabolite itaconate andfail to control C. burnetii replication in vitro and in vivo. Itaconate directly blocks C. burnetii replication at physiological concentrations. Finally, treatment with itaconate restored control of C. burnetii in murine and human macrophages and in vivo in mice. The work was performed in collaboration with the Lührmann group in a joint project in the CRC1181 Resolution of Inflammation and was supported by contributions from research groups from Erlangen, Jena, Regensburg, Munich, Düsseldorf and Osaka.
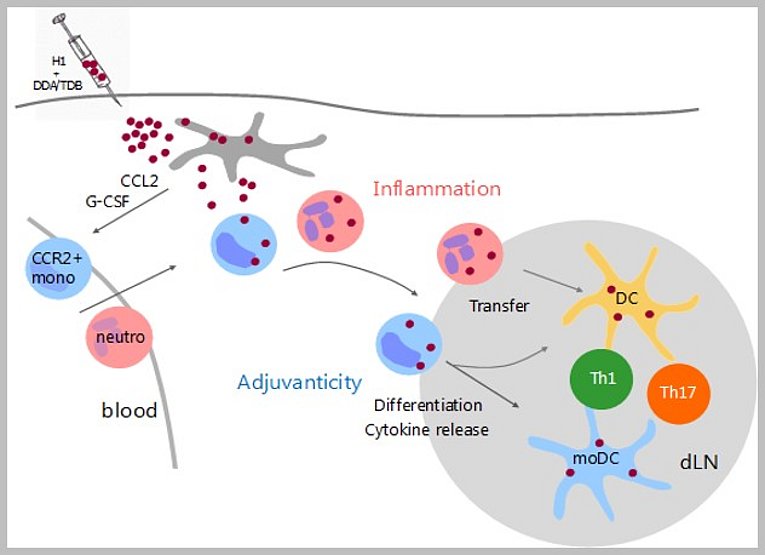
Understanding the cellular basis of a Th1/Th17-inducing adjuvant
Desel, C., Murray PJ, Lehmann CHK, Heger L, Christensen D, Anderse P, Mack M, Dudziak D, Lang R. Monocytes elicit a neutrophil-independent Th1/Th17 response upon immunization with a Mincle-dependent glycolipid adjuvant, Frontiers in Immunology 2022, 13:880474; doi: 10.3389/fimmu.2022.880474. 2022
https://www.frontiersin.org/articles/10.3389/fimmu.2022.880474/full
Development of subunit vaccines is hampered by a lack of adjuvants. The synthetic glycolipid TDB is a promising new adjuvant inducing protective Th1/Th17 immune responses via Mincle-Card9-dependent activation of antigen-presenting cells. The orchestration of Th1/Th17 induction by different innate immune cells is not well understood. Here, postdoctoral fellow Christiane Desel defined CCR2+ monocytes as the critical cell type for TDB-adjuvanticity, whereas neutrophils were dispensable. These findings provide a cellular mechanism for the generation of T cell responses, and furthermore suggest that unwanted side effects due to neutrophil activation may be dissociated from monocyte-elicited protective immunity.
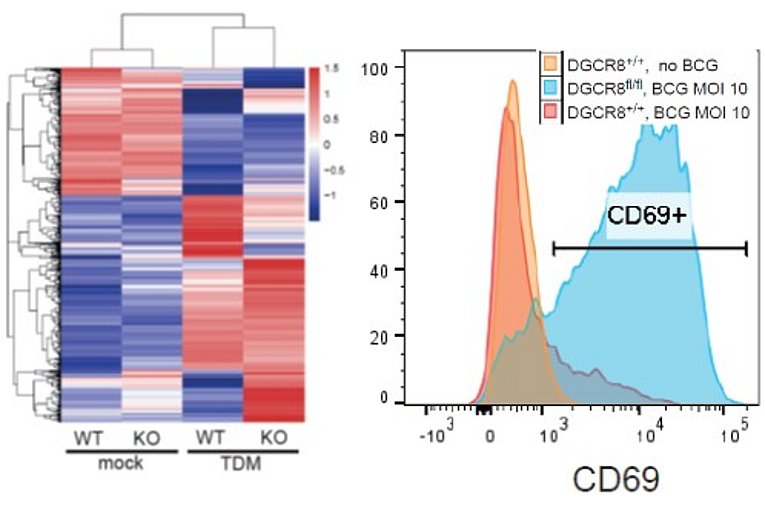
Lack of miRNAs in DGCR8-deficiency does not block macrophage differentiation but causes overshooting type I IFN response to mycobacteria
Killy B, Bodendorfer B, Mages J, Ritter K, Schreiber J, Hölscher C, Pracht K, Ekici A, Jäck HM, Lang R. DGCR8 deficiency impairs macrophage growth and unleashes the interferon response to mycobacteria, Life Science Alliance 2021, 4 (6): e202000810.
https://www.life-science-alliance.org/content/4/6/e202000810
MicroRNAs (miRNA) play an important role in regulation gene expression in innate immune cells and may be exploited by intracellular bacteria. In her PhD project, Barbara Killy addressed the question how a global abrogation of miRNA biosynthesis in DGCR8-deficient bone marrow cells affects the differentiation of macrophages and their interaction with the mycobacterial Mincle ligand TDM. Morphologically macrophages developed despite the lack of DGCR8, but were prone to overshooting type I interferon responses to TDM and to stimulation with whole bacteria of the Mycobacterium tuberculosis complex. Whether high IFNb expression in the absence of regulation by miRNA benefits the host or the mycobacteria will be interesting to explore in future studies.
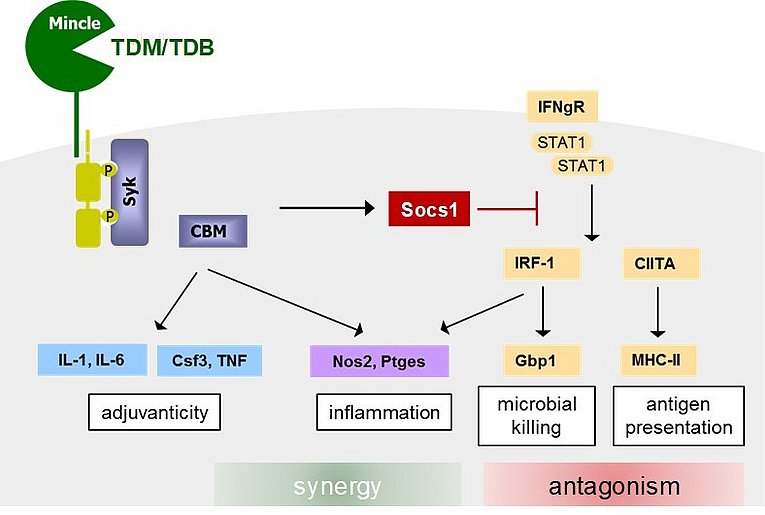
Antagonism and synergy in the macrophage response to IFNg and the mycobacterial cord factor
Huber A et al. Mycobacterial cord factor reprograms the macrophage response to IFNg towards enhanced inflammation yet impaired antigen presentation and expression of GBP1. J Immunol August 12, 2020, ji2000337; DOI: doi.org/10.4049/jimmunol.2000337
Trehalose-6,6-dimycolate (aka the cord factor) is the major PAMP of the mycobacterial cell wall and activates inflammatory cytokine production through MINCLE-CARD9 signaling. Alexandra Huber, Barbara Killy, Nadine Grummer and Barbara Bodendorfer, together with groups from Erlangen and Dresden show in this manuscript that cord factor signaling has ambiguous effects on the macrophage response to IFNg: while inflammatory cytokine and mediator production are enhanced, IFNg-induced MHC-II expression and induction of several GBPs is suppressed by the cord factor. This antagonism of IFNg-induced antigen presentation and anti-mycobacterial proteins may contribute to establish the intracellular niche of M. tuberculosis in its macrophage host.
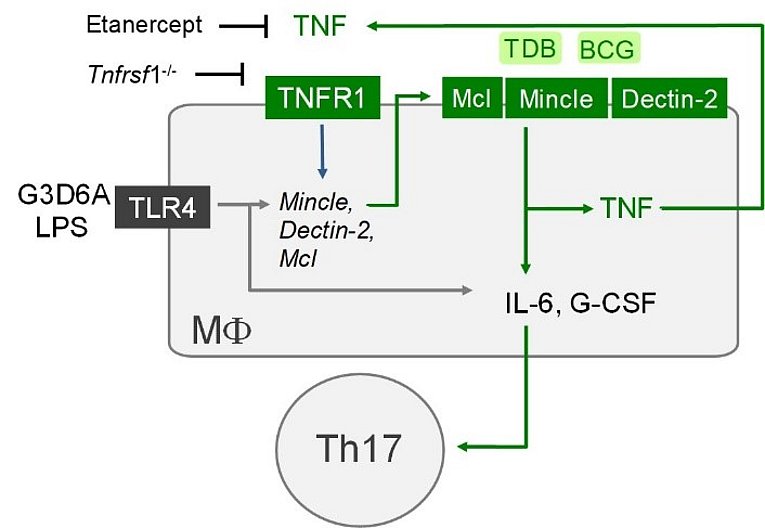
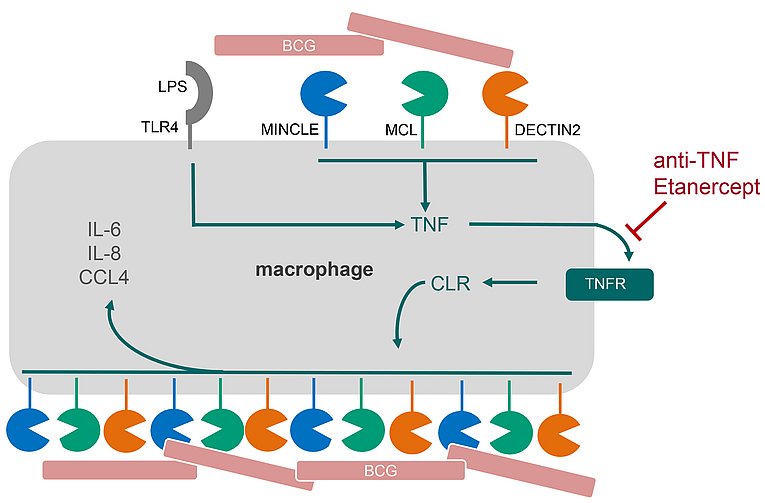
TNF is essential for MINCLE expression: a new function for an old cytokine in innate immunity
Schick J et al. TNF is essential for mycobacteria-induced MINCLE expression, macrophage activation and Th17 adjuvanticity.
J Immunol (Cutting Edge) July 15, 2020, 205 (2) 323-328; DOI: https://doi.org/10.4049/jimmunol.200042
Several C-type lectin receptors (CLR) involved in the recognition of mycobacteria are upregulated on macrophages by microbial stimuli. The work by Judith Schick and colleagues from Erlangen, Munich, Düsseldorf, Borstel and Copenhagen now demonstrates that upregulation of the cord factor-receptor MINCLE in macrophages depends on the cytokine TNF. In vivo, deletion or pharmacologic blockade of TNF prevented Th17 adjuvanticity of the synthetic MINCLE ligand TDB. The findings implicate regulation of MINCLE and related CLR as a potential new mechanism in inhibition of resistance to mycobacteria and in vaccination responses by TNF blockade. The later will be further investigated in a PhD project in the new GRK 2599 (see https://www.lymphozyten.med.fau.de/ for more information).
Next Generation Sequencing enhances detection of bacteria in polymicrobial CNS infection
Stebner A et al., (2020) Molecular diagnosis of polymicrobial brain abscesses with 16S-rDNA-based next-generation sequencing, Clinical Microbiology and Infection, doi.org/10.1016/j.cmi.2020.03.028
Bacterial brain abscesses are severe infections requiring prolonged antibiotic treatment. Identification of bacteria present in brain abscesses is important for appropriate treatment but difficult with standard microbiological methods. In a collaboration with the Institute of Virology and the Department of Neurosurgery, Alexander Stebner tested the feasibility of Illumina MiSeq Next Generation Sequencing of 16S rDNA amplicons in a large set of samples from patients with brain abscess or bacterial meningitis. The majority of brain abscesses was indeed polymicrobial, with up to 16 different bacteria taxa detected per sample. The results published in Clinical Microbiology and Infection demonstrate that NGS expands the spectrum of bacteria detected in brain abscesses and is suitable for metagenomic diagnostics of this severe infection
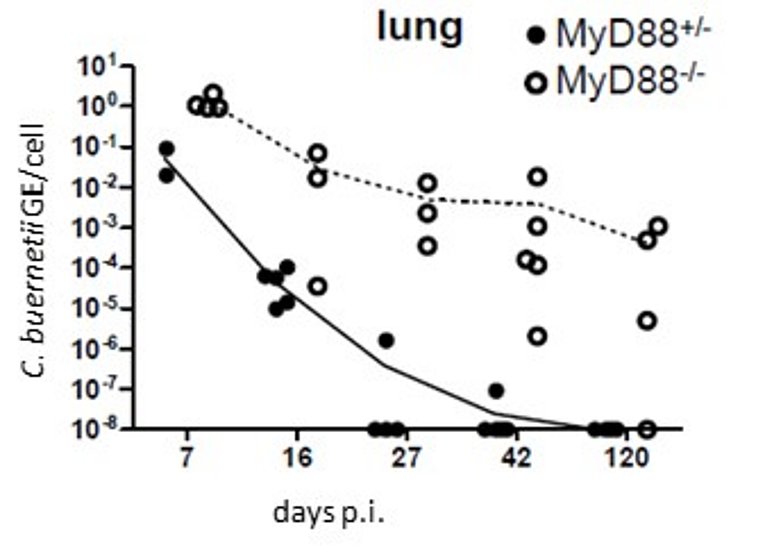
Persistent infection with Coxiella burnetii in MyD88-knockout mice: a new mouse model for chronic Q fever
Kohl L, Hayek I, Daniel C, Schulze-Lührmann J, Bodendorfer B, Lührmann A and Lang R (2019) MyD88 Is Required for Efficient Control of Coxiella burnetii Infection and Dissemination. Front. Immunol. 10:165. doi: 10.3389/fimmu.2019.00165
Infection with the zoonotic bacterium Coxiella burnetii is mostly self-limiting and resolves within several weeks, but becomes chronic and severe in some patients. To investigate the contribution of innate immune sensing mechanisms in the decision between resolution and chronicity, Lisa Kohl has used mice deficient in the central TLR adapter MyD88 in infection models with the attenuated Coxiella burnetii strain Nine Mile Phase II. Strongly increased bacterial burden, that persisted in the lungs for at least 4 months after infection, show that TLR signaling is essential for early control of C. burnetii replication and to prevent persistent chronic infection. The study was performed in collaboration with the lab of Anja Lührmann and with Dr. Christoph Daniel (Department of Nephropathology).
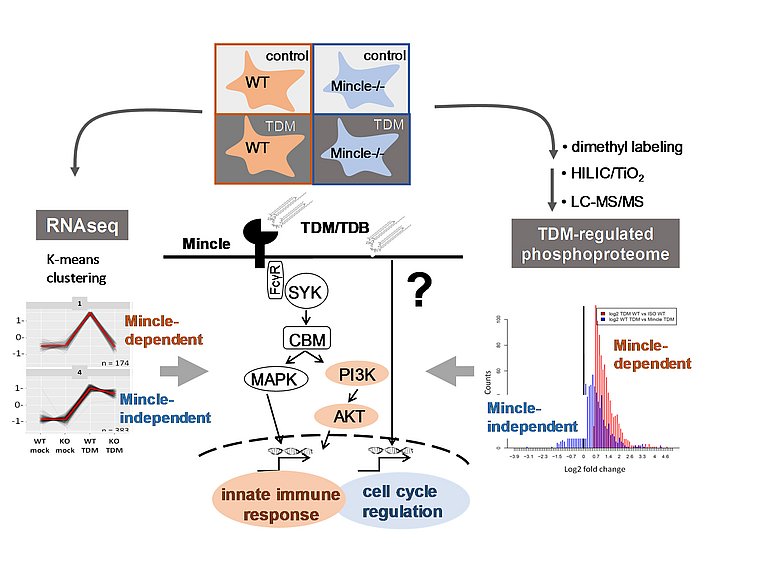
Global profiling of cord factor-induced signaling and gene expression in macrophages
Hansen M, Peltier J, et al. (2019) Macrophage Phosphoproteome Analysis Reveals MINCLE-dependent and –independent Mycobacterial Cord Factor Signaling Molecular & Cellular Proteomics 18: 669–685, 2019. DOI: 10.1074/mcp. RA118.000929.
In a collaboration with Julien Peltier from the lab of Matthias Trost, Madlen Hansen performed the first quantitative phosphoproteome analysis of TDM-activated macrophages. Surprisingly, the bioinformatic results revealed Mincle-dependent and -independent phosphorylation, which appear to affect different biological processes. Whereas PI3K/AKT signaling, dependent on Mincle, is involved in TDM-induced cytokine regulation, Mincle-independent phosphorylation and transcriptomic changes were linked to cell cycle regulation. Collectively, the combined phosphoproteome and RNAseq datasets show substantial reprogramming of macrophages by TDM and reveal a dichotomy of MINCLE-dependent and -independent signaling linked to distinct biological responses.
PUBLICATIONS
1. Zenebe, Y., M. Abebe, A. Munshea, G. Yismaw, M. Zewde, M. Alemayehu, R. Lang, and M. Habtamu. 2024. Intestinal Helminth Infections and Their Association with QuantiFERON-TB Gold Plus Test Performance in an Endemic Setting, Northwest Ethiopia. Infect Drug Resist 17: 4487-4500. https://doi.org/10.2147/IDR.S476492. PMC11492910
2. Siddique, M., F. Kellermeier, M. Olke, M. Zhao, K. Bussow, P. J. Oefner, A. Luhrmann, K. Dettmer, and R. Lang. 2024. Divergent effects of itaconate isomers on Coxiella burnetii growth in macrophages and in axenic culture. Front Immunol 15: 1427457. https://doi.org/10.3389/fimmu.2024.1427457. PMC11327005
3. Madzar, D., F. T. Nickel, V. Rothhammer, P. Goelitz, W. Geissdorfer, R. Dumke, and R. Lang. 2024. Meningitis and intracranial abscess due to Mycoplasma pneumoniae in a B cell-depleted patient with multiple sclerosis. Eur J Clin Microbiol Infect Dis 10.1007/s10096-024-04935-3: https://doi.org/10.1007/s10096-024-04935-3.
4. Lang, R., and M. Siddique. 2024. Control of immune cell signaling by the immuno-metabolite itaconate. Front Immunol 15: https://doi.org/10.3389/fimmu.2024.1352165.
5. Flores-Valdez, M. A., E. J. R. Peterson, M. J. Aceves-Sanchez, N. S. Baliga, Y. S. Morita, I. L. Sparks, D. K. Saini, R. Yadav, R. Lang, D. Mata-Espinosa, J. C. Leon-Contreras, and R. Hernandez-Pando. 2024. Comparison of the transcriptome, lipidome, and c-di-GMP production between BCGDeltaBCG1419c and BCG, with Mincle- and Myd88-dependent induction of proinflammatory cytokines in murine macrophages. Sci Rep 14: 11898. https://doi.org/10.1038/s41598-024-61815-8. PMC11126594
6. Dorner, H., I. Stolzer, J. Mattner, S. Kaminski, S. Leistl, L. M. Edrich, R. Schwendner, J. Hobauer, A. Sebald, S. Leikam, M. G. Acera, M. Dull, R. Lang, G. Seidel, T. Seitz, C. Hellerbrand, G. Fuhrmann, U. Distler, S. Tenzer, P. Eichhorn, M. Vieth, C. Schramm, P. Arnold, C. Becker, C. Weidinger, B. Siegmund, R. Atreya, M. Leppkes, E. Naschberger, F. Sampaziotis, P. Dietrich, M. Rauh, S. Wirtz, A. E. Kremer, M. F. Neurath, and C. Gunther. 2024. Gut Pathobiont-Derived Outer Membrane Vesicles Drive Liver Inflammation and Fibrosis in Primary Sclerosing Cholangitis-Associated Inflammatory Bowel Disease. Gastroenterology 10.1053/j.gastro.2024.06.032: https://doi.org/10.1053/j.gastro.2024.06.032.
7. Zenebe, Y., M. Habtamu, M. Abebe, B. Tulu, A. Atnafu, D. Mekonnen, R. Lang, and A. Munshea. 2023. Intestinal helminth co-infection and associated factors among pulmonary tuberculosis patients in Africa and Asia: a systematic review and meta-analysis. BMC Infect Dis 23: 739. https://doi.org/10.1186/s12879-023-08716-9. PMC10614413
8. Schick, J., M. Altunay, M. Lacorcia, N. Marschner, S. Westermann, J. Schluckebier, C. Schubart, B. Bodendorfer, D. Christensen, C. Alexander, S. Wirtz, D. Voehringer, C. P. da Costa, and R. Lang. 2023. IL-4 and helminth infection downregulate MINCLE-dependent macrophage response to mycobacteria and Th17 adjuvanticity. Elife 12: https://doi.org/10.7554/eLife.72923. PMC9908076
9. Kohl, L., M. Siddique, B. Bodendorfer, R. Berger, A. Preikschat, C. Daniel, M. Olke, E. Liebler-Tenorio, J. Schulze-Luehrmann, M. Mauermeir, K. T. Yang, I. Hayek, M. Szperlinski, J. Andrack, U. Schleicher, A. Bozec, G. Kronke, P. J. Murray, S. Wirtz, M. Yamamoto, V. Schatz, J. Jantsch, P. Oefner, D. Degrandi, K. Pfeffer, K. Mertens-Scholz, S. Rauber, C. Bogdan, K. Dettmer, A. Luhrmann, and R. Lang. 2023. Macrophages inhibit Coxiella burnetii by the ACOD1-itaconate pathway for containment of Q fever. EMBO Mol Med 15: e15931. https://doi.org/10.15252/emmm.202215931. PMC9906395
10. Knott, M., P. Hoelter, L. Soder, S. Schlaffer, S. Hoffmanns, R. Lang, A. Doerfler, and M. A. Schmidt. 2023. Can Perfusion-Based Brain Tissue Oxygenation MRI Support the Understanding of Cerebral Abscesses In Vivo? Diagnostics (Basel) 13: https://doi.org/10.3390/diagnostics13213346. PMC10647595
11. Haertl, T. O., D.; Schwinn, L.; Hirsch, C.; Eskofier, B.; Lang, R.; Wirtz, S.; Loos H. 2023. Exploring the interrelationship between the skin microbiome and skin volatiles: A pilot study. Front. Ecol. Evol. 11: https://doi.org/10.3389.
12. Haberkamp, C., I. Allabauer, N. Blaha, B. Bodendorfer, S. Cunningham, A. Hoerning, and R. Lang. 2023. TNF promotes DECTIN2 family C-type lectin receptor expression in human macrophages. J Leukoc Biol 113: 615-625. https://doi.org/10.1093/jleuko/qiad029.
13. Corbett, C., P. Finger, M. Heiss-Neumann, J. Bohnert, I. B. Eder, M. Eisele, I. Friesen, A. J. Kaasch, J. Kehrmann, R. Lang, J. Roedel, S. Roessler, A. Schmidt, S. Schneitler, D. Schui, F. Schuler, L. Sedlacek, A. Serr, A. G. Sitaru, J. Steinmann, D. Wagner, T. A. Wichelhaus, S. Hofmann-Thiel, and H. Hoffmann. 2023. Development of prevalence and incidence of non-tuberculous Mycobacteria in German laboratories from 2016 to 2020. Emerg Microbes Infect 10.1080/22221751.2023.2276342: 2276342. https://doi.org/10.1080/22221751.2023.2276342.
14. Blamberg, R., and R. Lang. 2023. The early bird catches the IL-2 in C-type lectin receptor-dependent activation of dendritic cells. Sci Signal 16: eadg4314. https://doi.org/10.1126/scisignal.adg4314.
15. Dorr, D., B. Obermayer, J. M. Weiner, K. Zimmermann, C. Anania, L. K. Wagner, E. M. Lyras, V. Sapozhnikova, D. Lara-Astiaso, F. Prosper, R. Lang, D. G. Lupianez, D. Beule, U. E. Hopken, A. Leutz, and A. Mildner. 2022. C/EBPbeta regulates lipid metabolism and Pparg isoform 2 expression in alveolar macrophages. Sci Immunol 7: eabj0140. https://doi.org/10.1126/sciimmunol.abj0140.
16. Dietschmann, A., S. Schruefer, S. Westermann, F. Henkel, K. Castiglione, R. Willebrand, J. Adam, J. Ruland, R. Lang, D. C. Sheppard, J. Esser-von-Bieren, D. Radtke, S. Krappmann, and D. Voehringer. 2022. Phosphatidylinositol 3-Kinase (PI3K) Orchestrates Aspergillus fumigatus-Induced Eosinophil Activation Independently of Canonical Toll-Like Receptor (TLR)/C-Type-Lectin Receptor (CLR) Signaling. mBio 10.1128/mbio.01239-22: e0123922. https://doi.org/10.1128/mbio.01239-22.
17. Desel, C., P. J. Murray, C. H. K. Lehmann, L. Heger, D. Christensen, P. Andersen, M. Mack, D. Dudziak, and R. Lang. 2022. Monocytes Elicit a Neutrophil-Independent Th1/Th17 Response Upon Immunization With a Mincle-Dependent Glycolipid Adjuvant. Front Immunol 13: 880474. https://doi.org/10.3389/fimmu.2022.880474. PMC9108773
18. Benard, A., M. J. Podolska, F. Czubayko, I. Kutschick, B. Klosch, A. Jacobsen, E. Naschberger, M. Brunner, C. Krautz, D. I. Trufa, H. Sirbu, R. Lang, R. Grutzmann, and G. F. Weber. 2022. Pleural Resident Macrophages and Pleural IRA B Cells Promote Efficient Immunity Against Pneumonia by Inducing Early Pleural Space Inflammation. Front Immunol 13: 821480. https://doi.org/10.3389/fimmu.2022.821480. PMC9047739
19. Stebner, A., A. Ensser, W. Geissdorfer, Y. Bozhkov, and R. Lang. 2021. Molecular diagnosis of polymicrobial brain abscesses with 16S-rDNA-based next-generation sequencing. Clin Microbiol Infect 27: 76-82. https://doi.org/10.1016/j.cmi.2020.03.028.
20. Killy, B., B. Bodendorfer, J. Mages, K. Ritter, J. Schreiber, C. Holscher, K. Pracht, A. Ekici, H. M. Jack, and R. Lang. 2021. DGCR8 deficiency impairs macrophage growth and unleashes the interferon response to mycobacteria. Life Sci Alliance 4: https://doi.org/10.26508/lsa.202000810. PMC8008949
21. Faas, M., N. Ipseiz, J. Ackermann, S. Culemann, A. Gruneboom, F. Schroder, T. Rothe, C. Scholtysek, M. Eberhardt, M. Bottcher, P. Kirchner, C. Stoll, A. Ekici, M. Fuchs, M. Kunz, B. Weigmann, S. Wirtz, R. Lang, J. Hofmann, J. Vera, D. Voehringer, A. Michelucci, D. Mougiakakos, S. Uderhardt, G. Schett, and G. Kronke. 2021. IL-33-induced metabolic reprogramming controls the differentiation of alternatively activated macrophages and the resolution of inflammation. Immunity 54: 2531-2546 e2535. https://doi.org/10.1016/j.immuni.2021.09.010.
22. Schick, J., J. Schafer, C. Alexander, S. Dichtl, P. J. Murray, D. Christensen, U. Sorg, K. Pfeffer, U. Schleicher, and R. Lang. 2020. Cutting Edge: TNF Is Essential for Mycobacteria-Induced MINCLE Expression, Macrophage Activation, and Th17 Adjuvanticity. J Immunol 205: 323-328. https://doi.org/10.4049/jimmunol.2000420.
23. Klessing, S., V. Temchura, P. Tannig, A. S. Peter, D. Christensen, R. Lang, and K. Uberla. 2020. CD4(+) T Cells Induced by Tuberculosis Subunit Vaccine H1 Can Improve the HIV-1 Env Humoral Response by Intrastructural Help. Vaccines (Basel) 8: https://doi.org/10.3390/vaccines8040604. PMC7711721
24. Huber, A., B. Killy, N. Grummel, B. Bodendorfer, S. Paul, V. Wiesmann, E. Naschberger, J. Zimmer, S. Wirtz, U. Schleicher, J. Vera, A. B. Ekici, A. Dalpke, and R. Lang. 2020. Mycobacterial Cord Factor Reprograms the Macrophage Response to IFN-gamma towards Enhanced Inflammation yet Impaired Antigen Presentation and Expression of GBP1. J Immunol 205: 1580-1592. https://doi.org/10.4049/jimmunol.2000337.
25. Andreev, D., M. Liu, D. Weidner, K. Kachler, M. Faas, A. Gruneboom, U. Schlotzer-Schrehardt, L. E. Munoz, U. Steffen, B. Grotsch, B. Killy, G. Kronke, A. M. Luebke, A. Niemeier, F. Wehrhan, R. Lang, G. Schett, and A. Bozec. 2020. Osteocyte necrosis triggers osteoclast-mediated bone loss through macrophage-inducible C-type lectin. J Clin Invest 130: 4811-4830. https://doi.org/10.1172/JCI134214. PMC7456234
26. Weerasekera, D., T. Fastner, R. Lang, A. Burkovski, and L. Ott. 2019. Of mice and men: Interaction of Corynebacterium diphtheriae strains with murine and human phagocytes. Virulence 10: 414-428. https://doi.org/10.1080/21505594.2019.1614384.
27. Pulido, R., and R. Lang. 2019. Dual Specificity Phosphatases: From Molecular Mechanisms to Biological Function. Int J Mol Sci 20: https://doi.org/10.3390/ijms20184372. 6770836
28. Moller, J., M. Kraner, U. Sonnewald, V. Sangal, H. Tittlbach, J. Winkler, T. H. Winkler, V. Melnikov, R. Lang, A. Sing, A. L. Mattos-Guaraldi, and A. Burkovski. 2019. Proteomics of diphtheria toxoid vaccines reveals multiple proteins that are immunogenic and may contribute to protection of humans against Corynebacterium diphtheriae. Vaccine 37: 3061-3070. https://doi.org/10.1016/j.vaccine.2019.04.059.
29. Lepenies, B., and R. Lang. 2019. Editorial: Lectins and Their Ligands in Shaping Immune Responses. Front Immunol 10: 2379. https://doi.org/10.3389/fimmu.2019.02379. 6794494
30. Lang, R., and F. A. M. Raffi. 2019. Dual-Specificity Phosphatases in Immunity and Infection: An Update. Int J Mol Sci 20: https://doi.org/10.3390/ijms20112710.
31. Kohl, L., I. Hayek, C. Daniel, J. Schulze-Luhrmann, B. Bodendorfer, A. Luhrmann, and R. Lang. 2019. MyD88 Is Required for Efficient Control of Coxiella burnetii Infection and Dissemination. Front Immunol 10: 165. https://doi.org/10.3389/fimmu.2019.00165. PMC6376249
32. Hayek, I., F. Fischer, J. Schulze-Luehrmann, K. Dettmer, K. Sobotta, V. Schatz, L. Kohl, K. Boden, R. Lang, P. J. Oefner, S. Wirtz, J. Jantsch, and A. Luhrmann. 2019. Limitation of TCA Cycle Intermediates Represents an Oxygen-Independent Nutritional Antibacterial Effector Mechanism of Macrophages. Cell Rep 26: 3502-3510 e3506. https://doi.org/10.1016/j.celrep.2019.02.103.
33. Hansen, M., J. Peltier, B. Killy, B. Amin, B. Bodendorfer, A. Hartlova, S. Uebel, M. Bosmann, J. Hofmann, C. Buttner, A. B. Ekici, M. Kuttke, H. Franzyk, C. Foged, S. Beer-Hammer, G. Schabbauer, M. Trost, and R. Lang. 2019. Macrophage Phosphoproteome Analysis Reveals MINCLE-dependent and -independent Mycobacterial Cord Factor Signaling. Mol Cell Proteomics 18: 669-685. https://doi.org/10.1074/mcp.RA118.000929. PMC6442366
34. Spitzer, P., R. Lang, T. J. Oberstein, P. Lewczuk, N. Ermann, H. B. Huttner, I. Masouris, J. Kornhuber, U. Kodel, and J. M. Maler. 2018. A Specific Reduction in Abeta1-42 vs. a Universal Loss of Abeta Peptides in CSF Differentiates Alzheimer's Disease From Meningitis and Multiple Sclerosis. Front Aging Neurosci 10: 152. https://doi.org/10.3389/fnagi.2018.00152. 5976781
35. Patzelt, T., S. J. Keppler, O. Gorka, S. Thoene, T. Wartewig, M. Reth, I. Forster, R. Lang, M. Buchner, and J. Ruland. 2018. Foxp1 controls mature B cell survival and the development of follicular and B-1 B cells. Proc Natl Acad Sci U S A 115: 3120-3125. https://doi.org/10.1073/pnas.1711335115. 5866538
36. Benard, A., I. Sakwa, P. Schierloh, A. Colom, I. Mercier, L. Tailleux, L. Jouneau, P. Boudinot, T. Al-Saati, R. Lang, J. Rehwinkel, A. G. Loxton, S. H. E. Kaufmann, V. Anton-Leberre, A. O'Garra, M. D. C. Sasiain, B. Gicquel, S. Fillatreau, O. Neyrolles, and D. Hudrisier. 2018. B Cells Producing Type I IFN Modulate Macrophage Polarization in Tuberculosis. Am J Respir Crit Care Med 197: 801-813. https://doi.org/10.1164/rccm.201707-1475OC. 5855072
37. Zega, K., V. M. Jovanovic, Z. Vitic, M. Niedzielska, L. Knaapi, M. M. Jukic, J. Partanen, R. F. Friedel, R. Lang, and C. Brodski. 2017. Dusp16 Deficiency Causes Congenital Obstructive Hydrocephalus and Brain Overgrowth by Expansion of the Neural Progenitor Pool. Front Mol Neurosci 10: 372. https://doi.org/10.3389/fnmol.2017.00372. 5684737
38. Winterholler, M., R. Coras, W. Geissdorfer, R. Rammensee, P. Golitz, C. Bogdan, and R. Lang. 2017. Fatal Mycotic Aneurysm of the Basilar Artery Caused by Aspergillus fumigatus in a Patient with Pituitary Adenoma and Meningitis. Front Med (Lausanne) 4: 113. https://doi.org/10.3389/fmed.2017.00113. 5513951
39. Velasquez, L. N., M. A. Milillo, M. V. Delpino, A. Trotta, P. Fernandez, R. G. Pozner, R. Lang, L. Balboa, G. H. Giambartolomei, and P. Barrionuevo. 2017. Brucella abortus down-regulates MHC class II by the IL-6-dependent inhibition of CIITA through the downmodulation of IFN regulatory factor-1 (IRF-1). J Leukoc Biol 101: 759-773. https://doi.org/10.1189/jlb.4A0416-196R.
40. Tima, H. G., J. R. Al Dulayymi, O. Denis, P. Lehebel, K. S. Baols, M. O. Mohammed, L. L'Homme, M. M. Sahb, G. Potemberg, S. Legrand, R. Lang, R. Beyaert, J. Piette, M. S. Baird, K. Huygen, and M. Romano. 2017. Inflammatory Properties and Adjuvant Potential of Synthetic Glycolipids Homologous to Mycolate Esters of the Cell Wall of Mycobacterium tuberculosis. J Innate Immun 9: 162-180. https://doi.org/10.1159/000450955.
41. Steininger, P. A., T. Bobinger, W. Dietrich, D. H. Lee, M. Knott, C. Bogdan, K. Korn, and R. Lang. 2017. Two Cases of Severe Tick-Borne Encephalitis in Rituximab-Treated Patients in Germany: Implications for Diagnosis and Prevention. Open Forum Infect Dis 4: ofx204. https://doi.org/10.1093/ofid/ofx204. 5903409
42. Schmok, E., M. Abad Dar, J. Behrends, H. Erdmann, D. Ruckerl, T. Endermann, L. Heitmann, M. Hessmann, A. Yoshimura, S. Rose-John, J. Scheller, U. E. Schaible, S. Ehlers, R. Lang, and C. Holscher. 2017. Suppressor of Cytokine Signaling 3 in Macrophages Prevents Exacerbated Interleukin-6-Dependent Arginase-1 Activity and Early Permissiveness to Experimental Tuberculosis. Front Immunol 8: 1537. https://doi.org/10.3389/fimmu.2017.01537. 5686055
43. Schick, J., P. Etschel, R. Bailo, L. Ott, A. Bhatt, B. Lepenies, C. Kirschning, A. Burkovski, and R. Lang. 2017. Toll-Like Receptor 2 and Mincle Cooperatively Sense Corynebacterial Cell Wall Glycolipids. Infect Immun 85: https://doi.org/10.1128/IAI.00075-17. PMC5478951
44. Rath, T., R. Atreya, W. Geissdorfer, R. Lang, A. Nagel, and M. F. Neurath. 2017. A Severe Case of Tuberculosis Radiologically and Endoscopically Mimicking Colorectal Cancer with Peritoneal Carcinomatosis. Case Rep Gastrointest Med 2017: 6206951. https://doi.org/10.1155/2017/6206951. 5646308
45. Patin, E. C., A. C. Geffken, S. Willcocks, C. Leschczyk, A. Haas, F. Nimmerjahn, R. Lang, T. H. Ward, and U. E. Schaible. 2017. Trehalose dimycolate interferes with FcgammaR-mediated phagosome maturation through Mincle, SHP-1 and FcgammaRIIB signalling. PLoS One 12: e0174973. https://doi.org/10.1371/journal.pone.0174973.
46. Ott, L., E. Hacker, T. Kunert, I. Karrington, P. Etschel, R. Lang, V. Wiesmann, T. Wittenberg, A. Singh, C. Varela, A. Bhatt, V. Sangal, and A. Burkovski. 2017. Analysis of Corynebacterium diphtheriae macrophage interaction: Dispensability of corynomycolic acids for inhibition of phagolysosome maturation and identification of a new gene involved in synthesis of the corynomycolic acid layer. PLoS One 12: e0180105. https://doi.org/10.1371/journal.pone.0180105.
47. Ostrop, J., and R. Lang. 2017. Contact, Collaboration, and Conflict: Signal Integration of Syk-Coupled C-Type Lectin Receptors. J Immunol 198: 1403-1414. https://doi.org/10.4049/jimmunol.1601665.
48. Lang, R., and J. Schick. 2017. Review: Impact of Helminth Infection on Antimycobacterial Immunity-A Focus on the Macrophage. Front Immunol 8: 1864. https://doi.org/10.3389/fimmu.2017.01864. 5743664
49. Lang, R., and J. Mattner. 2017. The role of lipids in host microbe interactions. Front Biosci (Landmark Ed) 22: 1581-1598. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/28410133.
50. Kao, D., A. Lux, A. Schaffert, R. Lang, F. Altmann, and F. Nimmerjahn. 2017. IgG subclass and vaccination stimulus determine changes in antigen specific antibody glycosylation in mice. Eur J Immunol 47: 2070-2079. https://doi.org/10.1002/eji.201747208.
51. Jurkin, J., C. Krump, R. Koffel, C. Fieber, C. Schuster, P. M. Brunner, I. Borek, G. Eisenwort, C. Lim, J. Mages, R. Lang, W. Bauer, D. Mechtcheriakova, A. Meshcheryakova, A. Elbe-Burger, G. Stingl, and H. Strobl. 2017. Human skin dendritic cell fate is differentially regulated by the monocyte identity factor Kruppel-like factor 4 during steady state and inflammation. J Allergy Clin Immunol 139: 1873-1884 e1810. https://doi.org/10.1016/j.jaci.2016.09.018.
52. Spitzer, P., M. Condic, M. Herrmann, T. J. Oberstein, M. Scharin-Mehlmann, D. F. Gilbert, O. Friedrich, T. Gromer, J. Kornhuber, R. Lang, and J. M. Maler. 2016. Amyloidogenic amyloid-beta-peptide variants induce microbial agglutination and exert antimicrobial activity. Sci Rep 6: 32228. https://doi.org/10.1038/srep32228.
53. Patin, E. C., S. Willcocks, S. Orr, T. H. Ward, R. Lang, and U. E. Schaible. 2016. Mincle-mediated anti-inflammatory IL-10 response counter-regulates IL-12 in vitro. Innate Immun 22: 181-185. https://doi.org/10.1177/1753425916636671. 4826952
54. Patin, E. C., A. V. Jones, A. Thompson, M. Clement, C. T. Liao, J. S. Griffiths, L. E. Wallace, C. E. Bryant, R. Lang, P. Rosenstiel, I. R. Humphreys, P. R. Taylor, G. W. Jones, and S. J. Orr. 2016. IL-27 Induced by Select Candida spp. via TLR7/NOD2 Signaling and IFN-beta Production Inhibits Fungal Clearance. J Immunol 197: 208-221. https://doi.org/10.4049/jimmunol.1501204. 4911616
55. Maor-Nof, M., E. Romi, H. S. Shalom, V. Ulisse, C. Raanan, A. Nof, D. Leshkowitz, R. Lang, and A. Yaron. 2016. Axonal Degeneration Is Regulated by a Transcriptional Program that Coordinates Expression of Pro- and Anti-degenerative Factors. Neuron 92: 991-1006. https://doi.org/10.1016/j.neuron.2016.10.061.
56. Hupfer, T., J. Schick, K. Jozefowski, D. Voehringer, J. Ostrop, and R. Lang. 2016. Stat6-Dependent Inhibition of Mincle Expression in Mouse and Human Antigen-Presenting Cells by the Th2 Cytokine IL-4. Front Immunol 7: 423. https://doi.org/10.3389/fimmu.2016.00423. 5063851
57. Huber, A., R. S. Kallerup, K. S. Korsholm, H. Franzyk, B. Lepenies, D. Christensen, C. Foged, and R. Lang. 2016. Trehalose diester glycolipids are superior to the monoesters in binding to Mincle, activation of macrophages in vitro and adjuvant activity in vivo. Innate Immun 22: 405-418. https://doi.org/10.1177/1753425916651132.
58. Wenzel, J., J. L. Ouderkirk, M. Krendel, and R. Lang. 2015. Class I myosin Myo1e regulates TLR4-triggered macrophage spreading, chemokine release, and antigen presentation via MHC class II. Eur J Immunol 45: 225-237. https://doi.org/10.1002/eji.201444698.
59. Siegert, I., J. Schodel, M. Nairz, V. Schatz, K. Dettmer, C. Dick, J. Kalucka, K. Franke, M. Ehrenschwender, G. Schley, A. Beneke, J. Sutter, M. Moll, C. Hellerbrand, B. Wielockx, D. M. Katschinski, R. Lang, B. Galy, M. W. Hentze, P. Koivunen, P. J. Oefner, C. Bogdan, G. Weiss, C. Willam, and J. Jantsch. 2015. Ferritin-Mediated Iron Sequestration Stabilizes Hypoxia-Inducible Factor-1alpha upon LPS Activation in the Presence of Ample Oxygen. Cell Rep 13: 2048-2055. https://doi.org/10.1016/j.celrep.2015.11.005.
60. Rabes, A., S. Zimmermann, K. Reppe, R. Lang, P. H. Seeberger, N. Suttorp, M. Witzenrath, B. Lepenies, and B. Opitz. 2015. The C-Type Lectin Receptor Mincle Binds to Streptococcus pneumoniae but Plays a Limited Role in the Anti-Pneumococcal Innate Immune Response. Plos One 10: https://doi.org/ARTN e0117022; 10.1371/journal.pone.0117022.
61. Ostrop, J., K. Jozefowski, S. Zimmermann, K. Hofmann, E. Strasser, B. Lepenies, and R. Lang. 2015. Contribution of MINCLE-SYK Signaling to Activation of Primary Human APCs by Mycobacterial Cord Factor and the Novel Adjuvant TDB. J Immunol 195: 2417-2428. https://doi.org/10.4049/jimmunol.1500102.
62. Niedzielska, M., F. A. Raffi, J. Tel, S. Muench, K. Jozefowski, N. Alati, K. Lahl, J. Mages, U. Billmeier, M. Schiemann, U. K. Appelt, S. Wirtz, T. Sparwasser, H. Hochrein, C. G. Figdor, S. M. Keyse, and R. Lang. 2015. Selective Expression of the MAPK Phosphatase Dusp9/MKP-4 in Mouse Plasmacytoid Dendritic Cells and Regulation of IFN-beta Production. J Immunol 195: 1753-1762. https://doi.org/10.4049/jimmunol.1400658.
63. Goldmann, T., N. Zeller, J. Raasch, K. Kierdorf, K. Frenzel, L. Ketscher, A. Basters, O. Staszewski, S. M. Brendecke, A. Spiess, T. L. Tay, C. Kreutz, J. Timmer, G. M. Mancini, T. Blank, G. Fritz, K. Biber, R. Lang, D. Malo, D. Merkler, M. Heikenwalder, K. P. Knobeloch, and M. Prinz. 2015. USP18 lack in microglia causes destructive interferonopathy of the mouse brain. EMBO J 10.15252/embj.201490791: https://doi.org/10.15252/embj.201490791.
64. Schoenen, H., A. Huber, N. Sonda, S. Zimmermann, J. Jantsch, B. Lepenies, V. Bronte, and R. Lang. 2014. Differential control of Mincle-dependent cord factor recognition and macrophage responses by the transcription factors C/EBPbeta and HIF1alpha. J Immunol 193: 3664-3675. https://doi.org/10.4049/jimmunol.1301593.
65. Niedzielska, M., B. Bodendorfer, S. Munch, A. Eichner, M. Derigs, O. da Costa, A. Schweizer, F. Neff, L. Nitschke, T. Sparwasser, S. M. Keyse, and R. Lang. 2014. Gene trap mice reveal an essential function of dual specificity phosphatase Dusp16/MKP-7 in perinatal survival and regulation of Toll-like receptor (TLR)-induced cytokine production. J Biol Chem 289: 2112-2126. https://doi.org/10.1074/jbc.M113.535245. 3900958
66. Goktuna, S. I., O. Canli, J. Bollrath, A. A. Fingerle, D. Horst, M. A. Diamanti, C. Pallangyo, M. Bennecke, T. Nebelsiek, A. K. Mankan, R. Lang, D. Artis, Y. Hu, T. Patzelt, J. Ruland, T. Kirchner, M. M. Taketo, A. Chariot, M. C. Arkan, and F. R. Greten. 2014. IKKalpha promotes intestinal tumorigenesis by limiting recruitment of M1-like polarized myeloid cells. Cell Rep 7: 1914-1925. https://doi.org/10.1016/j.celrep.2014.05.006.
67. Globisch, T., N. Steiner, L. Fulle, V. Lukacs-Kornek, D. Degrandi, P. Dresing, J. Alferink, R. Lang, K. Pfeffer, M. Beyer, H. Weighardt, C. Kurts, T. Ulas, J. L. Schultze, and I. Forster. 2014. Cytokine-dependent regulation of dendritic cell differentiation in the splenic microenvironment. Eur J Immunol 44: 500-510. https://doi.org/10.1002/eji.201343820.
68. Friedrich, O., B. Yi, J. N. Edwards, B. Reischl, A. Wirth-Hucking, A. Buttgereit, R. Lang, C. Weber, F. Polyak, I. Liu, F. von Wegner, T. R. Cully, A. Lee, P. Most, and M. Volkers. 2014. IL-1alpha reversibly inhibits skeletal muscle ryanodine receptor. a novel mechanism for critical illness myopathy? Am J Respir Cell Mol Biol 50: 1096-1106. https://doi.org/10.1165/rcmb.2013-0059OC. 4068910
69. Schwitalla, S., A. A. Fingerle, P. Cammareri, T. Nebelsiek, S. I. Goktuna, P. K. Ziegler, O. Canli, J. Heijmans, D. J. Huels, G. Moreaux, R. A. Rupec, M. Gerhard, R. Schmid, N. Barker, H. Clevers, R. Lang, J. Neumann, T. Kirchner, M. M. Taketo, G. R. van den Brink, O. J. Sansom, M. C. Arkan, and F. R. Greten. 2013. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell 152: 25-38. https://doi.org/10.1016/j.cell.2012.12.012.
70. Puttur, F., C. Arnold-Schrauf, K. Lahl, G. Solmaz, M. Lindenberg, C. T. Mayer, M. Gohmert, M. Swallow, C. van Helt, H. Schmitt, L. Nitschke, B. N. Lambrecht, R. Lang, M. Messerle, and T. Sparwasser. 2013. Absence of Siglec-H in MCMV infection elevates interferon alpha production but does not enhance viral clearance. PLoS Pathog 9: e1003648. https://doi.org/10.1371/journal.ppat.1003648. 3784486
71. Held, C., J. Wenzel, V. Wiesmann, R. Palmisano, R. Lang, and T. Wittenberg. 2013. Enhancing automated micrograph-based evaluation of LPS-stimulated macrophage spreading. Cytometry A 83: 409-418. https://doi.org/10.1002/cyto.a.22248.
72. Heitmann, L., H. Schoenen, S. Ehlers, R. Lang, and C. Holscher. 2013. Mincle is not essential for controlling Mycobacterium tuberculosis infection. Immunobiology 218: 506-516. https://doi.org/10.1016/j.imbio.2012.06.005.
73. Desel, C., K. Werninghaus, M. Ritter, K. Jozefowski, J. Wenzel, N. Russkamp, U. Schleicher, D. Christensen, S. Wirtz, C. Kirschning, E. M. Agger, C. P. da Costa, and R. Lang. 2013. The Mincle-Activating Adjuvant TDB Induces MyD88-Dependent Th1 and Th17 Responses through IL-1R Signaling. Plos One 8: https://doi.org/ARTN e53531
10.1371/journal.pone.0053531.
74. Specht, S., M. D. Taylor, M. A. Hoeve, J. E. Allen, R. Lang, and A. Hoerauf. 2012. Over expression of IL-10 by macrophages overcomes resistance to murine filariasis. Exp Parasitol 132: 90-96. https://doi.org/10.1016/j.exppara.2011.09.003.
75. Oeder, S., R. A. Jorres, I. Weichenmeier, G. Pusch, W. Schober, F. Pfab, H. Behrendt, R. Schierl, A. Kronseder, D. Nowak, S. Dietrich, E. Fernandez-Caldas, J. Lintelmann, R. Zimmermann, R. Lang, J. Mages, H. Fromme, and J. T. Buters. 2012. Airborne indoor particles from schools are more toxic than outdoor particles. Am J Respir Cell Mol Biol 47: 575-582. https://doi.org/10.1165/rcmb.2012-0139OC.
76. Frankenberger, M., T. P. Hofer, A. Marei, F. Dayyani, S. Schewe, C. Strasser, A. Aldraihim, F. Stanzel, R. Lang, R. Hoffmann, O. Prazeres da Costa, T. Buch, and L. Ziegler-Heitbrock. 2012. Transcript profiling of CD16-positive monocytes reveals a unique molecular fingerprint. Eur J Immunol 42: 957-974. https://doi.org/10.1002/eji.201141907.
77. Wenzel, J., C. Held, R. Palmisano, S. Teufel, J. P. David, T. Wittenberg, and R. Lang. 2011. Measurement of TLR-Induced Macrophage Spreading by Automated Image Analysis: Differential Role of Myd88 and MAPK in Early and Late Responses. Front Physiol 2: 71. https://doi.org/10.3389/fphys.2011.00071. 3198511
78. Strebovsky, J., P. Walker, R. Lang, and A. H. Dalpke. 2011. Suppressor of cytokine signaling 1 (SOCS1) limits NF kappa B signaling by decreasing p65 stability within the cell nucleus. Faseb Journal 25: 863-874. https://doi.org/10.1096/fj.10-170597.
79. Lang, R., H. Schoenen, and C. Desel. 2011. Targeting Syk-Card9-activating C-type lectin receptors by vaccine adjuvants: findings, implications and open questions. Immunobiology 216: 1184-1191. https://doi.org/10.1016/j.imbio.2011.06.005.
80. Kratochvill, F., C. Machacek, C. Vogl, F. Ebner, V. Sedlyarov, A. R. Gruber, H. Hartweger, R. Vielnascher, M. Karaghiosoff, T. Rulicke, M. Muller, I. Hofacker, R. Lang, and P. Kovarik. 2011. Tristetraprolin-driven regulatory circuit controls quality and timing of mRNA decay in inflammation. Mol Syst Biol 7: 560. https://doi.org/10.1038/msb.2011.93. 3737733
81. Klotz, C., T. Ziegler, A. S. Figueiredo, S. Rausch, M. R. Hepworth, N. Obsivac, C. Sers, R. Lang, P. Hammerstein, R. Lucius, and S. Hartmann. 2011. A helminth immunomodulator exploits host signaling events to regulate cytokine production in macrophages. PLoS Pathog 7: e1001248. https://doi.org/10.1371/journal.ppat.1001248. 3017123
82. Jantsch, J., M. Wiese, J. Schodel, K. Castiglione, J. Glasner, S. Kolbe, D. Mole, U. Schleicher, K. U. Eckardt, M. Hensel, R. Lang, C. Bogdan, M. Schnare, and C. Willam. 2011. Toll-like receptor activation and hypoxia use distinct signaling pathways to stabilize hypoxia-inducible factor 1 alpha (HIF1A) and result in differential HIF1A-dependent gene expression. Journal of Leukocyte Biology 90: 551-562. https://doi.org/10.1189/jlb.1210683.
83. Held, C., J. Wenzel, R. Webel, M. Marschall, R. Lang, R. Palmisano, and T. Wittenberg. 2011. Using multimodal information for the segmentation of fluorescent micrographs with application to virology and microbiology. Conf Proc IEEE Eng Med Biol Soc 2011: 6487-6490. https://doi.org/10.1109/IEMBS.2011.6091601.
84. Weintz, G., J. V. Olsen, K. Fruhauf, M. Niedzielska, I. Amit, J. Jantsch, J. Mages, C. Frech, L. Dolken, M. Mann, and R. Lang. 2010. The phosphoproteome of toll-like receptor-activated macrophages. Mol Syst Biol 6: 371. https://doi.org/10.1038/msb.2010.29. 2913394
85. Schoenen, H., B. Bodendorfer, K. Hitchens, S. Manzanero, K. Werninghaus, F. Nimmerjahn, E. M. Agger, S. Stenger, P. Andersen, J. Ruland, G. D. Brown, C. Wells, and R. Lang. 2010. Cutting edge: Mincle is essential for recognition and adjuvanticity of the mycobacterial cord factor and its synthetic analog trehalose-dibehenate. J Immunol 184: 2756-2760. https://doi.org/10.4049/jimmunol.0904013. 3442336
86. Rodriguez, N., H. Dietrich, I. Mossbrugger, G. Weintz, J. Scheller, M. Hammer, L. Quintanilla-Martinez, S. Rose-John, T. Miethke, and R. Lang. 2010. Increased inflammation and impaired resistance to Chlamydophila pneumoniae infection in Dusp1(-/-) mice: critical role of IL-6. J Leukoc Biol 88: 579-587. https://doi.org/10.1189/jlb.0210083.
87. Layland, L. E., J. Mages, C. Loddenkemper, A. Hoerauf, H. Wagner, R. Lang, and C. U. P. da Costa. 2010. Pronounced Phenotype in Activated Regulatory T Cells during a Chronic Helminth Infection. Journal of Immunology 184: 713-724. https://doi.org/10.4049/jimmunol.0901435.
88. Ingersoll, M. A., R. Spanbroek, C. Lottaz, E. L. Gautier, M. Frankenberger, R. Hoffmann, R. Lang, M. Haniffa, M. Collin, F. Tacke, A. J. R. Habenicht, L. Ziegler-Heitbrock, and G. J. Randolph. 2010. Comparison of gene expression profiles between human and mouse monocyte subsets. (vol 115, pg e10, 2010). Blood 116: 857-857. https://doi.org/, <Go to ISI>://WOS:000280596500032.
89. Hammer, M., B. Echtenachter, H. Weighardt, K. Jozefowski, S. Rose-John, D. N. Mannel, B. Holzmann, and R. Lang. 2010. Increased inflammation and lethality of Dusp1-/- mice in polymicrobial peritonitis models. Immunology 131: 395-404. https://doi.org/10.1111/j.1365-2567.2010.03313.x. 2996560
90. Werninghaus, K., A. Babiak, O. Gross, C. Holscher, H. Dietrich, E. M. Agger, J. Mages, A. Mocsai, H. Schoenen, K. Finger, F. Nimmerjahn, G. D. Brown, C. Kirschning, A. Heit, P. Andersen, H. Wagner, J. Ruland, and R. Lang. 2009. Adjuvanticity of a synthetic cord factor analogue for subunit Mycobacterium tuberculosis vaccination requires FcRgamma-Syk-Card9-dependent innate immune activation. J Exp Med 206: 89-97. https://doi.org/10.1084/jem.20081445. 2626670
91. Schreiber, T., S. Ehlers, L. Heitmann, A. Rausch, J. Mages, P. J. Murray, R. Lang, and C. Holscher. 2009. Autocrine IL-10 induces hallmarks of alternative activation in macrophages and suppresses antituberculosis effector mechanisms without compromising T cell immunity. J Immunol 183: 1301-1312. https://doi.org/10.4049/jimmunol.0803567. 2735238
92. Schaljo, B., F. Kratochvill, N. Gratz, I. Sadzak, I. Sauer, M. Hammer, C. Vogl, B. Strobl, M. Muller, P. J. Blackshear, V. Poli, R. Lang, P. J. Murray, and P. Kovarik. 2009. Tristetraprolin is required for full anti-inflammatory response of murine macrophages to IL-10. J Immunol 183: 1197-1206. https://doi.org/10.4049/jimmunol.0803883. 2755621
93. Renstrom, J., R. Istvanffy, K. Gauthier, A. Shimono, J. Mages, A. Jardon-Alvarez, M. Kroger, M. Schiemann, D. H. Busch, I. Esposito, R. Lang, C. Peschel, and R. A. Oostendorp. 2009. Secreted frizzled-related protein 1 extrinsically regulates cycling activity and maintenance of hematopoietic stem cells. Cell Stem Cell 5: 157-167. https://doi.org/10.1016/j.stem.2009.05.020.
94. Rad, R., W. Ballhorn, P. Voland, K. Eisenacher, J. Mages, L. Rad, R. Ferstl, R. Lang, H. Wagner, R. M. Schmid, S. Bauer, C. Prinz, C. J. Kirschning, and A. Krug. 2009. Extracellular and intracellular pattern recognition receptors cooperate in the recognition of Helicobacter pylori. Gastroenterology 136: 2247-2257. https://doi.org/10.1053/j.gastro.2009.02.066.
95. Hertle, M. L., C. Popp, S. Petermann, S. Maier, E. Kremmer, R. Lang, J. Mages, and B. Kempkes. 2009. Differential gene expression patterns of EBV infected EBNA-3A positive and negative human B lymphocytes. PLoS Pathog 5: e1000506. https://doi.org/10.1371/journal.ppat.1000506. 2700271
96. Andersen, C. A., I. Rosenkrands, A. W. Olsen, P. Nordly, D. Christensen, R. Lang, C. Kirschning, J. M. Gomes, V. Bhowruth, D. E. Minnikin, G. S. Besra, F. Follmann, P. Andersen, and E. M. Agger. 2009. Novel generation mycobacterial adjuvant based on liposome-encapsulated monomycoloyl glycerol from Mycobacterium bovis bacillus Calmette-Guerin. J Immunol 183: 2294-2302. https://doi.org/10.4049/jimmunol.0804091.
97. Aly, S., J. Mages, N. Reiling, U. Kalinke, T. Decker, R. Lang, and S. Ehlers. 2009. Mycobacteria-induced granuloma necrosis depends on IRF-1. Journal of Cellular and Molecular Medicine 13: 2069-2082. https://doi.org/10.1111/j.1582-4934.2008.00470.x.
98. von Burstin, J., S. Eser, B. Seidler, A. Meining, M. Bajbouj, J. Mages, R. Lang, A. J. Kind, A. E. Schnieke, R. M. Schmid, G. Schneider, and D. Saur. 2008. Highly sensitive detection of early-stage pancreatic cancer by multimodal near-infrared molecular imaging in living mice. International Journal of Cancer 123: 2138-2147. https://doi.org/10.1002/ijc.23780.
99. Rodriguez, N., R. Lang, N. Wantia, C. Cirl, T. Ertl, S. Durr, H. Wagner, and T. Miethke. 2008. Induction of iNOS by Chlamydophila pneumoniae requires MyD88-dependent activation of JNK. Journal of Leukocyte Biology 84: 1585-1593. https://doi.org/10.1189/jlb.0508304.
100. Prinz, M., H. Schmidt, A. Mildner, K. P. Knobeloch, U. K. Hanisch, J. Raasch, D. Merkler, C. Detje, I. Gutcher, J. Mages, R. Lang, R. Martin, R. Gold, B. Becher, W. Bruck, and U. Kalinke. 2008. Distinct and nonredundant in vivo functions of IFNAR on myeloid cells limit autoimmunity in the central nervous system. Immunity 28: 675-686. https://doi.org/10.1016/j.immuni.2008.03.011.
101. Mages, J., K. Freimuller, R. Lang, A. K. Hatzopoulos, S. Guggemoos, U. H. Koszinowski, and H. Adler. 2008. Proteins of the secretory pathway govern virus productivity during lytic gammaherpesvirus infection. Journal of Cellular and Molecular Medicine 12: 1974-1989. https://doi.org/10.1111/j.1582-4934.2008.00235.x.
102. Ledran, M. H., A. Krassowska, L. Armstrong, I. Dimmick, J. Renstrom, R. Lang, S. Yung, M. Santibanez-Coref, E. Dzierzak, M. Stojkovic, R. A. J. Oostendorp, L. Forrester, and M. Lako. 2008. Efficient hematopoietic differentiation of human embryonic stem cells on stromal cells derived from hematopoietic niches. Cell Stem Cell 3: 85-98. https://doi.org/10.1016/j.stem.2008.06.001.
103. Kalali, B. N., G. Kollisch, J. Mages, T. Muller, S. Bauer, H. Wagner, J. Ring, R. Lang, M. Mempel, and M. Ollert. 2008. Double-stranded RNA induces an antiviral defense status in epidermal keratinocytes through TLR3-, PKR-, and MDA5/RIG-I-mediated differential signaling. J Immunol 181: 2694-2704. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/18684960.
104. Hofer, T. P. J., M. Frankenberger, J. Mages, R. Lang, R. Hoffmann, A. Colige, and L. Ziegler-Heitbrock. 2008. Tissue-specific induction of ADAMTS2 in monocytes and macrophages by glucocorticoids. Journal of Molecular Medicine-Jmm 86: 323-332. https://doi.org/10.1007/s00109-007-0284-0.
105. Farhat, K., S. Riekenberg, H. Heine, J. Debarry, R. Lang, J. Mages, U. Buwitt-Beckmann, K. Roeschmann, G. Jung, K. H. Wiesmuller, and A. J. Ulmer. 2008. Heterodimerization of TLR2 with TLR1 or TLR6 expands the ligand spectrum but does not lead to differential signaling. Journal of Leukocyte Biology 83: 692-701. https://doi.org/10.1189/jlb.0807586.
106. Eckmann, L., T. Nebelsiek, A. A. Fingerle, S. M. Dann, J. Mages, R. Lang, S. Robine, M. F. Kagnoff, R. M. Schmid, M. Karin, M. C. Arkan, and F. R. Greten. 2008. Opposing functions of IKK beta during acute and chronic intestinal inflammation. Proceedings of the National Academy of Sciences of the United States of America 105: 15058-15063. https://doi.org/10.1073/pnas.0808216105.
107. Agger, E. M., I. Rosenkrands, J. Hansen, K. Brahimi, B. S. Vandahl, C. Aagaard, K. Werninghaus, C. Kirschning, R. Lang, D. Christensen, M. Theisen, F. Follmann, and P. Andersen. 2008. Cationic liposomes formulated with synthetic mycobacterial cordfactor (CAF01): a versatile adjuvant for vaccines with different immunological requirements. PLoS One 3: e3116. https://doi.org/10.1371/journal.pone.0003116. 2525815
108. Schmitz, F., A. Heit, S. Guggemoos, A. Krug, J. Mages, M. Schiemann, H. Adler, I. Drexler, T. Haas, R. Lang, and H. Wagner. 2007. Interferon-regulatory-factor 1 controls Toll-like receptor 9-mediated IFN-beta production in myeloid dendritic cells. Eur J Immunol 37: 315-327. https://doi.org/10.1002/eji.200636767.
109. Rodriguez, N., J. Mages, H. Dietrich, N. Wantia, H. Wagner, R. Lang, and T. Miethke. 2007. MyD88-dependent changes in the pulmonary transcriptome after infection with Chlamydia pneumoniae. Physiol Genomics 30: 134-145. https://doi.org/10.1152/physiolgenomics.00011.2007.
110. Rad, R., L. Brenner, A. Krug, P. Voland, J. Mages, R. Lang, S. Schwendy, W. Reindl, A. Dossumbekova, W. Ballhorn, H. Wagner, R. M. Schmid, S. Bauer, and C. Prinz. 2007. Toll-like receptor-dependent activation of antigen-presenting cells affects adaptive immunity to Helicobacter pylori. Gastroenterology 133: 150-163 e153. https://doi.org/10.1053/j.gastro.2007.04.071.
111. Oeder, S., J. Mages, P. Flicek, and R. Lang. 2007. Uncovering information on expression of natural antisense transcripts in Affymetrix MOE430 datasets. BMC Genomics 8: 200. https://doi.org/10.1186/1471-2164-8-200. 1929078
112. Mayer, A. K., M. Muehmer, J. Mages, K. Gueinzius, C. Hess, K. Heeg, R. Bals, R. Lang, and A. H. Dalpke. 2007. Differential recognition of TLR-dependent microbial ligands in human bronchial epithelial cells. J Immunol 178: 3134-3142. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/17312161.
113. Mages, J., H. Dietrich, and R. Lang. 2007. A genome-wide analysis of LPS tolerance in macrophages. Immunobiology 212: 723-737. https://doi.org/10.1016/j.imbio.2007.09.015.
114. Aly, S., T. Laskay, J. Mages, A. Malzan, R. Lang, and S. Ehlers. 2007. Interferon-gamma-dependent mechanisms of mycobacteria-induced pulmonary immunopathology: the role of angiostasis and CXCR3-targeted chemokines for granuloma necrosis. J Pathol 212: 295-305. https://doi.org/10.1002/path.2185.
115. Weighardt, H., J. Mages, G. Jusek, S. Kaiser-Moore, R. Lang, and B. Holzmann. 2006. Organ-specific role of MyD88 for gene regulation during polymicrobial peritonitis. Infect Immun 74: 3618-3632. https://doi.org/10.1128/IAI.01681-05. 1479251
116. Maier, S., G. Staffler, A. Hartmann, J. Hock, K. Henning, K. Grabusic, R. Mailhammer, R. Hoffmann, M. Wilmanns, R. Lang, J. Mages, and B. Kempkes. 2006. Cellular target genes of Epstein-Barr virus nuclear antigen 2. Journal of Virology 80: 9761-9771. https://doi.org/10.1128/Jvi.00665-06.
117. Lang, R., M. Hammer, and J. Mages. 2006. DUSP meet immunology: dual specificity MAPK phosphatases in control of the inflammatory response. J Immunol 177: 7497-7504. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/17114416.
118. Keller, C., R. Hoffmann, R. Lang, S. Brandau, C. Hermann, and S. Ehlers. 2006. Genetically determined susceptibility to tuberculosis in mice causally involves accelerated and enhanced recruitment of granulocytes. Infect Immun 74: 4295-4309. https://doi.org/10.1128/IAI.00057-06. 1489748
119. Hammer, M., J. Mages, H. Dietrich, A. Servatius, N. Howells, A. C. Cato, and R. Lang. 2006. Dual specificity phosphatase 1 (DUSP1) regulates a subset of LPS-induced genes and protects mice from lethal endotoxin shock. J Exp Med 203: 15-20. https://doi.org/10.1084/jem.20051753. 2118077
120. Dossumbekova, A., C. Prinz, J. Mages, R. Lang, J. G. Kusters, A. H. M. Van Vliet, W. Reindl, S. Backert, D. Saur, R. M. Schmid, and R. Rad. 2006. Helicobacter pylori HopH (OipA) and bacterial pathogenicity: Genetic and functional genomic analysis of hopH gene Polymorphisms. Journal of Infectious Diseases 194: 1346-1355. https://doi.org/Doi 10.1086/508426.
121. Ludwig, H., J. Mages, C. Staib, M. H. Lehmann, R. Lang, and G. Sutter. 2005. Role of viral factor E3L in modified vaccinia virus Ankara infection of human HeLa cells: Regulation of the virus life cycle and identification of differentially expressed host genes. Journal of Virology 79: 2584-2596. https://doi.org/10.1128/Jvi.79.4.2584-2596.2005.
122. Lang, R. 2005. Tuning of macrophage responses by Stat3-inducing cytokines: molecular mechanisms and consequences in infection. Immunobiology 210: 63-76. https://doi.org/10.1016/j.imbio.2005.05.001.
123. Hammer, M., J. Mages, H. Dietrich, F. Schmitz, F. Striebel, P. J. Murray, H. Wagner, and R. Lang. 2005. Control of dual-specificity phosphatase-1 expression in activated macrophages by IL-10. Eur J Immunol 35: 2991-3001. https://doi.org/10.1002/eji.200526192.
124. Weighardt, H., G. Jusek, J. Mages, R. Lang, K. Hoebe, B. Beutler, and B. Holzmann. 2004. Identification of a TLR4- and TRIF-dependent activation program of dendritic cells. European Journal of Immunology 34: 558-564. https://doi.org/10.1002/eji.200324714.
125. Schmitz, F., J. Mages, A. Heit, R. Lang, and H. Wagner. 2004. Transcriptional activation induced in macrophages by Toll-like receptor (TLR) ligands: from expression profiling to a model of TLR signaling. Eur J Immunol 34: 2863-2873. https://doi.org/10.1002/eji.200425228.
126. Rad, R., A. Dossumbekova, B. Neu, R. Lang, S. Bauer, D. Saur, M. Gerhard, and C. Prinz. 2004. Cytokine gene polymorphisms influence mucosal cytokine expression, gastric inflammation, and host specific colonisation during Helicobacter pylori infection. Gut 53: 1082-1089. https://doi.org/10.1136/gut.2003.029736.
127. Pauleau, A. L., R. Rutschman, R. Lang, A. Pernis, S. S. Watowich, and P. J. Murray. 2004. Enhancer-mediated control of macrophage-specific arginase I expression. J Immunol 172: 7565-7573. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/15187136.
128. Lang, R., A. L. Pauleau, E. Parganas, Y. Takahashi, J. Mages, J. N. Ihle, R. Rutschman, and P. J. Murray. 2003. SOCS3 regulates the plasticity of gp130 signaling. Nat Immunol 4: 546-550. https://doi.org/10.1038/ni932.
129. Rad, R., M. Gerhard, R. Lang, M. Schoniger, T. Rosch, W. Schepp, I. Becker, H. Wagner, and C. Prinz. 2002. The Helicobacter pylori blood group antigen-binding adhesin facilitates bacterial colonization and augments a nonspecific immune response. Journal of Immunology 168: 3033-3041. https://doi.org/, <Go to ISI>://WOS:000174280400058.
130. Lang, R., R. L. Rutschman, D. R. Greaves, and P. J. Murray. 2002. Autocrine deactivation of macrophages in transgenic mice constitutively overexpressing IL-10 under control of the human CD68 promoter. J Immunol 168: 3402-3411. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/11907098.
131. Lang, R., D. Patel, J. J. Morris, R. L. Rutschman, and P. J. Murray. 2002. Shaping gene expression in activated and resting primary macrophages by IL-10. J Immunol 169: 2253-2263. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/12193690.
132. Kornhuber, M. E., C. Ganz, R. Lang, T. Brill, and W. Schmahl. 2002. Focal encephalitis in the Lewis rat induced by intracerebral enterotoxin superantigen and amplified by activated intravenous splenocytes. Neuroscience Letters 324: 93-96. https://doi.org/Pii S0304-3940(02)00107-6; Doi 10.1016/S0304-3940(02)00107-6.
133. Rutschman, R., R. Lang, M. Hesse, J. N. Ihle, T. A. Wynn, and P. J. Murray. 2001. Cutting edge: Stat6-dependent substrate depletion regulates nitric oxide production. J Immunol 166: 2173-2177. https://doi.org/, http://www.ncbi.nlm.nih.gov/pubmed/11160269.
134. Kranzer, K., M. Bauer, G. B. Lipford, K. Heeg, H. Wagner, and R. Lang. 2000. CpG-oligodeoxynucleotides enhance T-cell receptor-triggered interferon-gamma production and up-regulation of CD69 via induction of antigen-presenting cell-derived interferon type I and interleukin-12. Immunology 99: 170-178. https://doi.org/DOI 10.1046/j.1365-2567.2000.00964.x.
135. Lang, R., L. Hultner, G. B. Lipford, H. Wagner, and K. Heeg. 1999. Guanosine-rich oligodeoxynucleotides induce proliferation of macrophage progenitors in cultures of murine bone marrow cells. European Journal of Immunology 29: 3496-3506. https://doi.org/Doi 10.1002/(Sici)1521-4141(199911)29:11<3496::Aid-Immu3496>3.3.Co;2-V.
136. Lang, R., and K. Heeg. 1998. Semiquantitative determination of human cytokine mRna expression using TaqMan RT-PCR. Inflammopharmacology 6: 297-309. https://doi.org/10.1007/s10787-998-0014-4.
137. Lang, R., K. Pfeffer, H. Wagner, and K. Heeg. 1997. A rapid method for semiquantitative analysis of the human V beta-repertoire using TaqMan(R) PCR. Journal of Immunological Methods 203: 181-192. https://doi.org/Doi 10.1016/S0022-1759(97)00028-8.
138. Lang, R., H. Wagner, and K. Heeg. 1995. Differential-Effects of the Immunosuppressive Agents Cyclosporine and Leflunomide in-Vivo - Leflunomide Blocks Clonal T-Cell Expansion yet Allows Production of Lymphokines and Manifestation of T-Cell-Mediated Shock. Transplantation 59: 382-389. https://doi.org/Doi 10.1097/00007890-199502150-00013.