Research group of Dr. C. Schwartz
Innate checkpoints of T cell regulation
Cells of the innate immune system initiate the adaptive immune response to infections and inflammatory stimuli. In order to efficiently mount a tailored T cell response three signals are required: MHC:TCR stimulation, co-stimulation and cytokines. Dendritic cells, macrophages and innate lymphoid cells are fully equipped to provide all three signals. Our research focuses on the cell-specific role of costimulatory molecules with activating and inhibiting function - and what regulates their expression.
One of these molecules is programmed death ligand 1 (PD-L1, CD274, B7-H1), which interacts with programmed cell death protein 1 (PD-1, CD279) on activated T cells. Classically, this protein-protein-interaction leads to the inhibition of the T cell response. Blockade with so-called immune checkpoint inhibitors re-activates T cells. In the context of Th1- and Th17-responses, such as cancer and autoimmunity, its inhibitory function is very well studied but the knowledge on the impact on other T cell subsets remains incomplete.
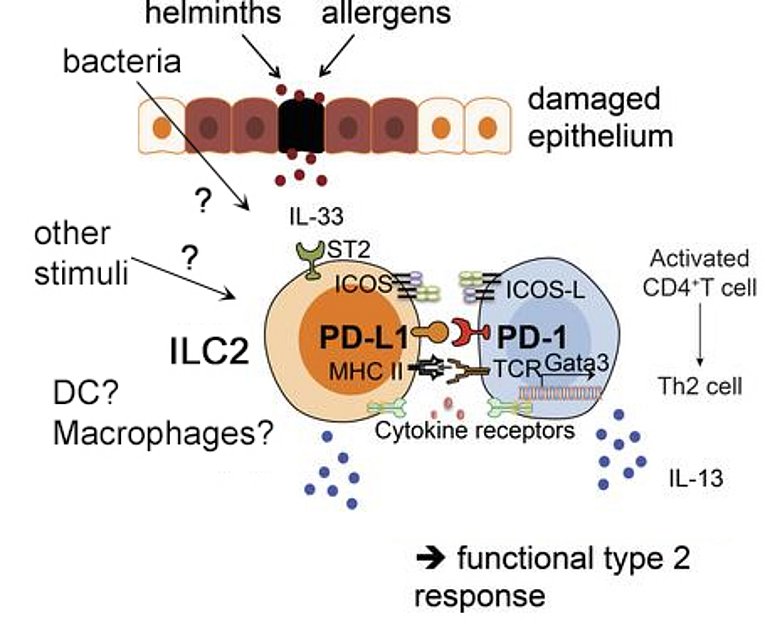
- PD-L1-mediated activation of type 2 responses. We have shown that PD-L1 can serve an activating function in the context of a type 2 polarised immune response against the helminth parasite Nippostrongylus brasiliensis (Figure 1). Expression of PD-L1 on pulmonary ILC2 activated Th2 cells and promoted expulsion of the parasite (Schwartz, 2017). However, it is currently unclear how PD-L1-expression on ILC2 influences T cell responses in other inflammatory situations. Furthermore, other innate cells, such as dendritic cells and macrophages express PD-L1 and shape the T cell immune response. Using a combination of in vivo and in vitro approaches in preclinical models together with patient samples, we study the non-classical function innate PD-L1 has on adaptive immunity.
- Bacterial regulation of co-stimulation. We recently reported an important role of the microbiota in the development of allergic inflammation and the activation of innate immune cells (Schwartz, 2019). Expression of costimulatory molecules on innate cells is dynamic and influenced by microbial stimuli. We will investigate how bacterial products, such as TLR ligands and metabolites, regulate PD-L1 expression on innate cells. The composition of the microbiome determines the availability of certain metabolites and thereby influences immune responses at distal sites. To this end, we will analyse the microbiome under steady state and inflammatory conditions to study the impact on PD-L1-mediated co-stimulation of T cell responses.